Home / Power Supplies /
How does a battery work?
The electric current is a flow of electrons that flow through a conductor cable. Electrons are negatively charged, and like two magnets that we want to put together, negative side with negative side or positive side with positive side, they repel.
This means that an electron repel another electron because they are negatively charged. Then, a positive charge will attract a negative charge, such as electrons.
Electric batteries, by means of a chemical reaction, produce:
- At its negative terminal, a lot of electrons (negatively charged) and …
- At its positive terminal, a lack of electrons (negatively charged).
Now, if this battery powers a circuit, a flow of electrons will leave the negative terminal of the battery, (because electrons repel each other and also repel the free electrons in the conductor), and are directed to the positive terminal, where there is a lack of electrons passing through the circuit to which it is connected. This is the way the electric current is created.
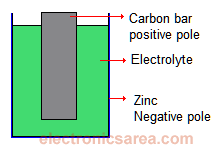
The chemical process does not occur indefinitely, and after some time it has no effect. (You can notice it because the battery voltage decreases). This is why the battery life is finite. The best known battery, is dry battery. See the diagram. By means of a chemical reaction:
- The zinc cover attracts electrons and becomes negatively charged.
- The carbon loses electrons and becomes positively charged.
Because the chemical reaction oxidizes the zinc, the battery has a limited life.
The common dry cell is the zinc–carbon battery, sometimes called the dry Leclanché cell, with a voltage of 1.5 volts.
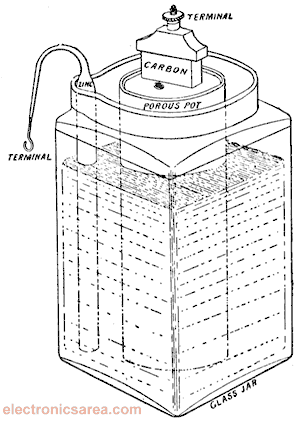
See page for author [Public domain], via Wikimedia Commons