Aug 032015
Home / Power Supplies /
Types of Cells / Batteries
Primary batteries (non-rechargeable)
1. Dry Batteries: Zinc Battery, Alkaline Battery. These batteries can not be recharged, and they discharge even when they are not in use. There are tubular cells of 1.5 volts and batteries built with layers. For example, the 9-volt battery is composed of several cells (layers) of 1.5 volts in series.
2. Mercury Batteries
- Have a longer life than dry batteries, and the voltage is more stable.
- They are smaller.
- They are used in hearing aids and compact electronics devices.
3. Alkaline battery
- It has a long life due to its concentration of energy.
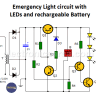
Secondary batteries (rechargeable)
- Nickel-cadmium alkaline battery. They are compact, can supply enough power, and have more stable voltage than the common batteries.
- Lead Battery. Widely used in the automotive industry
There are cells that are connected in series and are called batteries. Example: use automotive batteries. The main parameters of a cell or battery are:
- Voltage across its terminals
- Amp/hour Capacity
Cells or Batteries interconnection
- If two cells or batteries are connected in series, the current is the same for all and the total voltage is the sum of the voltages of the individual batteries or batteries
- If two cells or batteries are placed in parallel, they have the same voltage but higher current capacity.
Note: To interconnect cells or batteries in parallel, it is necessary that all batteries have the same voltage. Never interconnect new batteries with used batteries.
You may also like: How does a battery work?