Home / Capacitors /
The Electrolytic Capacitor
Electrolytic capacitors have been developed to achieve large capacities in small physical dimensions. To achieve this large capacity, a special dielectric is used.
The capacity of a capacitor (capacitance) can be obtained with the following formula: C = EA/d, where:
– A = plate’s surface in square meters (m2)
– d = separation between plates in meters (m)
– E = dielectric constant (Absolute permittivity) = 8.84 x 10-12
– C = Capacitance in Farads
For example:
If a plate’s surface is A = 0.15 m2 and the separation between plates is d = 6 x 103 m, What is the capacitance?
C = EA/d = (8.84 x 10-12)(0.15) / 6 x 103 = 0.221 x 10-15 F = 0.221nF (nanoFarads)
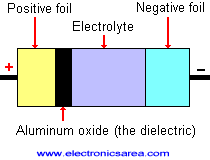
The bigger the dielectric constant (E), the bigger the capacitance will be. The electrolytic capacitor is made of two aluminum foils, separated by an absorbent paper impregnated with electrolyte.
An electric current is forced to pass through the plates of the capacitor, generating a chemical reaction. This chemical reaction creates a layer of aluminum oxide (the dielectric). Look at the picture.
Physically, the electrolytic capacitor is located inside a closed aluminum tube. It has a safety valve that opens when the electrolyte boils avoiding the risk of explosion.
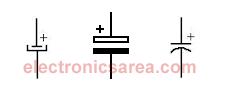
The electrolytic capacitors have a voltage polarity requirement, so their terminals could not be reversed. Usually the correct polarity is indicated on the body of the capacitor. One disadvantage of the electrolytic capacitor is that the maximum voltage allowed on its terminals is not very high.
When the capacitor is damaged, it is necessary to find another one with the same capacity and at least the same voltage (the same or higher), but … it is not recommended using a capacitor with a voltage (manufacturing data) well above the damaged one, because a capacitor that is used with a voltage much lower than the voltage for which it was designed, causes that the layer of aluminum oxide (the dielectric) decreases until the capacitor fails.
Note: This type of capacitors must be used as soon as possible after their manufacturing.
An electrolytic capacitor must not have a long storage period. If this kind of capacitor does not receive a voltage, it begins to harm itself. (The layer of aluminum oxide begins to decrease). It’s a good idea to take into account the manufacture’s date.