The Structure of Matter
Matter is divided into molecules, which are then divided into atoms. These atoms are composed of two parts: the nucleus and the periphery.
- The nucleus of an atom contains positively charged protons and neutrons, which, as their name suggests, have no electric charge.
- Negatively charged electrons are on the periphery of the atom.
The Bohr Atom
Danish physicist Niels Bohr created a model showing the structure of the atom, which was later called the Bohr model. See the diagram below.
Since the number of electrons (in blue) equals the number of protons (in red), the atom is said to be electrically neutral. The number of protons equals the number of electrons.
Some electrons are in the outer orbits of the atom and are the farthest from the core. These electrons can more easily escape the attraction of the nucleus and are called valence electrons.
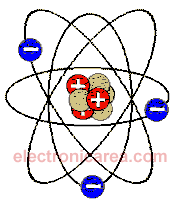
Example: The copper atom has 29 protons and 29 electrons. Twenty-eight of these electrons are in orbits close to the nucleus, and one is in a distant orbit. This electron is called a free or valence electron.
- A material with many free electrons is called a conductive material.
If a material has few free electrons, then it is an insulator.
Examples:
- Conductive materials include gold, silver, aluminum, and copper.
- Insulating materials include ceramics, glass, wood, and paper.
An atom with fewer electrons than normal is called a positive ion. An atom with more than normal number of electrons is called a negative ion.
Electricity
Electricity is the accumulation or movement of electrons that have been removed from their orbits. (See the picture above.)
These electrons are called free electrons and can easily move through a material when they are taken out of their orbits. This movement is called an electric current.